Paul W. Armstrong, MD @CVC_UAlberta @UAlberta @MerkCanada @Merk #Cardiovascular #HeartFailure #Research U.S. FDA App...
Paul W. Armstrong, MD Canadian VIGOUR Centre, University of Alberta, Edmonton, AB, Canada speaks about Merck Announces U.S. FDA Approval of VERQUVO® (vericiguat).
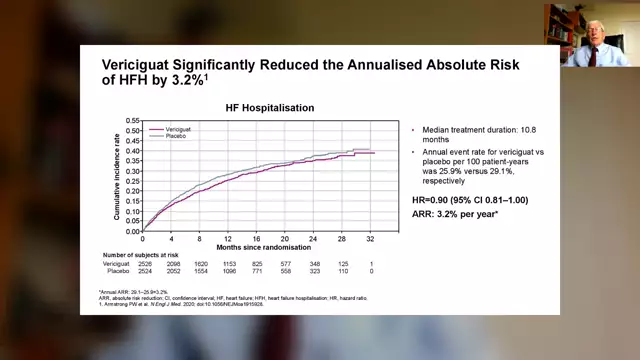
The primary efficacy objective of VICTORIA was to evaluate if VERQUVO, in conjunction with other heart failure therapies, is superior to placebo in reducing the risk of cardiovascular mortality or hospitalization for heart failure in adults with symptomatic recurrent heart failure and less than 45% of the ejection fraction following a worsening heart failure case. Based on a time-to-event study, VERQUVO reached the primary efficacy objective (hazard ratio [HR]: 0.90, 95 percent confidence interval [CI], 0.82-0.98; p=0.019). There was a 4.2 percent reduction in annualized absolute risk with VERQUVO compared to placebo over the course of the study. Therefore, to avoid one primary endpoint case, 24 patients will need to be monitored for an average of one year.
Information Supporting the Approval
VERQUVO approval was based on evidence from VICTORIA (NCT02861534), a randomized, concurrent, placebo-controlled, double-blind, event-driven, multi-center clinical trial comparing VERQUVO with placebo in 5,050 adult patients with New York Heart Association (NYHA) class II-IV symptomatic chronic heart failure and left ventricular ejection fraction (LVEF) less than 45% after a wound of symptomatic chronic heart failure (NYHA) class II-IV. A worsening case of heart failure was described as hospitalization for heart failure within six months or less prior to randomization or use for heart failure of outpatient IV diuretics within three months or less prior to randomization. In VICTORIA, the primary outcome was time-to-first cardiovascular death or heart failure hospitalization. The median follow-up time was 11 months for the primary endpoint. Based on a time-to-event analysis, VERQUVO was superior to placebo in reducing the risk of cardiovascular death or hospitalization for heart failure.
Patients were given VERQUVO 10 mg once daily or a matched placebo up to the target maintenance dose. Therapy started with VERQUVO 2.5 mg once daily and increased to 5 mg once daily and then 10 mg once daily, as tolerated, at roughly two-week intervals. The placebo doses were adjusted similarly. 90 percent of patients in both the VERQUVO and placebo arms were treated with the 10 mg target maintenance dose after approximately one year.
Participants in the study were: 76% male, 64% Caucasian, 22% Asian and 5% Black. The mean age was 67. 59 percent of patients were NYHA Class II at randomization, 40 percent were NYHA Class III and 1 percent were NYHA Class IV. The mean LVEF was 29 percent . Approximately half of all patients had an EF of less than 30%, and 14% of patients had an EF of between 40% and 45%. Sixty-seven percent of VICTORIA patients were enrolled within three months of a hospitalization index case for heart failure; 17 percent were enrolled within three to six months of hospitalization for heart failure; and 16 percent were enrolled within three months of outpatient care for worsening heart failure with IV diuretics. At randomization, the median NT-pro B-type natriuretic peptide (NT-proBNP) level was 2800 pg/mL.
Participants in the study were on quality of treatment. At baseline, 93 percent of patients received a beta-blocker, 73 percent received an angiotensin-converting enzyme (ACE) or angiotensin II receptor blocker (ARB) inhibitor, 70 percent received a mineralocorticoid receptor antagonist (MRA), 15 percent received an angiotensin receptor and neprilysin inhibitor (ARNI) combination, 28 percent had an implantable cardiac receptor antagonist (MRA), and 28 percent had an implantable cardiac inhibitor (ARNI). Ninety-one percent of patients were treated with two or more drugs for heart failure (beta-blocker, any receptor of the renin-angiotensin system [RAS], or MRA) and all three were treated in 60 percent of patients. At baseline, 6% of patients received an inhibitor of ivabradine and 3% of sodium glucose co-transporter 2 (SGLT2).
VERQUVO showed an adverse effect profile comparable to placebo in the VICTORIA trial. Hypotension (16% vs 15%) and anemia were the adverse drug reactions that occurred more frequently with VERQUVO than with placebo and in more than or equivalent to 5% of patients treated with VERQUVO in VICTORIA (10 percent vs 7 percent ). The VICTORIA trial included a total of 2,519 patients treated with VERQUVO (up to 10 mg once daily). The mean period of exposure to VERQUVO was one year, with a median duration of 2.6 years.